Author: kaye valila
Infections in Immunosuppressed Patients: Unusual Organisms and Risks
Explore the unique risks and unusual pathogens that affect immunosuppressed patients. Learn why these infections are silent and how doctors detect them.
Keep ReadingBiosimilar Cost Savings: Comparing Prices to Original Biologics
Compare the cost savings of biosimilars versus original biologic drugs. Learn why prices differ from generics and how to maximize savings in a complex market.
Keep ReadingHow to Measure Education Effectiveness and Track Patient Understanding
Learn how to measure education effectiveness in patient care using direct and indirect assessments, formative and summative tools to track genuine understanding.
Keep ReadingClinical Trial Eligibility: How Biomarkers are Changing Cancer Care
Learn how biomarkers are transforming clinical trial eligibility in cancer care, moving from broad categories to precision medicine and increasing drug success rates.
Keep ReadingMultiple Sclerosis Guide: How the Immune System Attacks Your Nerves
Learn how Multiple Sclerosis affects the nervous system, the role of the immune system in demyelination, and the latest treatments to slow disease progression.
Keep ReadingDeficiency Letters in Generic Applications: Common FDA Findings and How to Avoid Them
Common FDA deficiency letter findings in generic drug applications include dissolution method flaws, impurity control gaps, and inadequate drug substance characterization. Learn how to avoid these top issues and improve first-cycle approval rates.
Keep ReadingHow to Talk to Patients About Generic Medications: A Practical Guide for Providers
Learn how to effectively communicate with patients about generic medications to improve adherence, reduce costs, and build trust. Evidence-based strategies for providers to address common concerns and ensure better health outcomes.
Keep ReadingHow Medicare Drug Price Negotiations Work and What It Means for Insurer Discounts
Medicare’s new drug price negotiation program is cutting costs for 10 top medications by up to 79%, starting in 2026. Learn how this affects Medicare beneficiaries, private insurers, and what’s coming next.
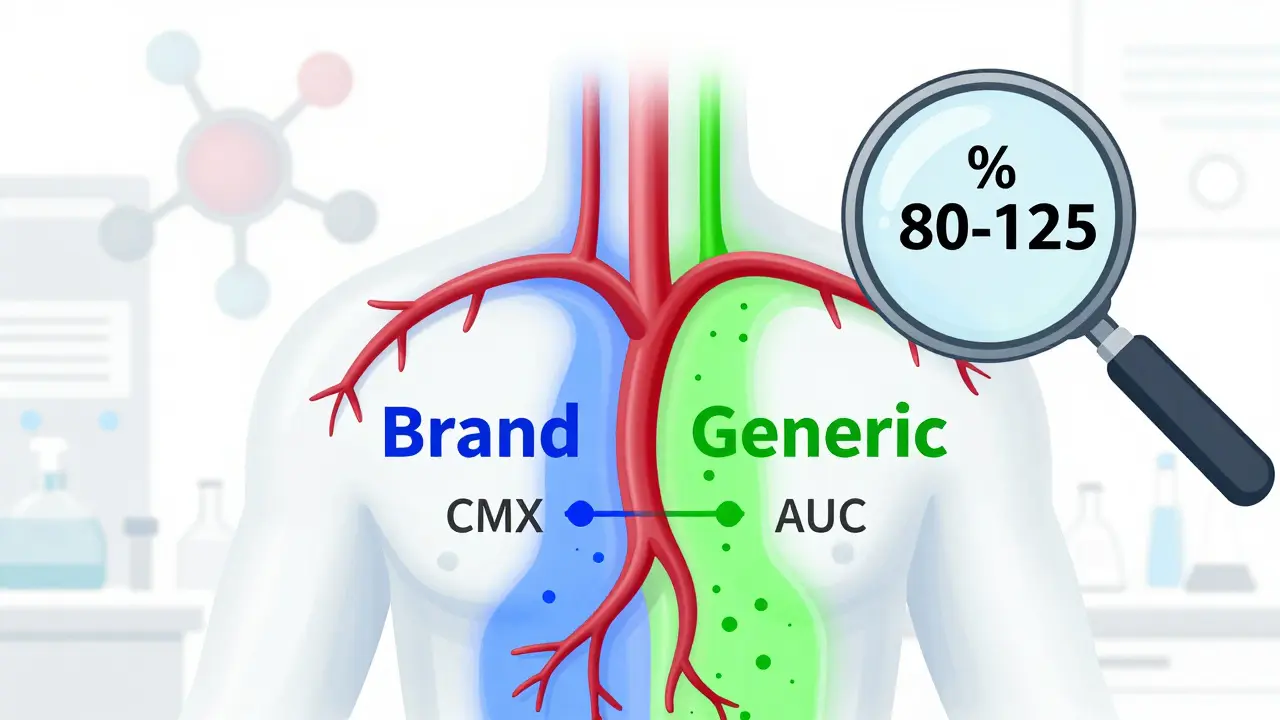
Keep ReadingPharmacokinetic Studies: The Real Standard for Generic Drug Equivalence
Pharmacokinetic studies are the primary method used to prove generic drugs are equivalent to brand-name versions, measuring how quickly and how much of the drug enters the bloodstream. But they’re not foolproof - especially for complex or topical drugs. Learn how these tests work, where they fall short, and what’s coming next.
Keep ReadingPregnancy Registries: What We’re Learning About Medication Safety
Pregnancy registries are collecting real-world data on medication safety during pregnancy, helping doctors and patients make informed choices. Here's what we're learning-and where the gaps still remain.
Keep Reading